Metabolic stability
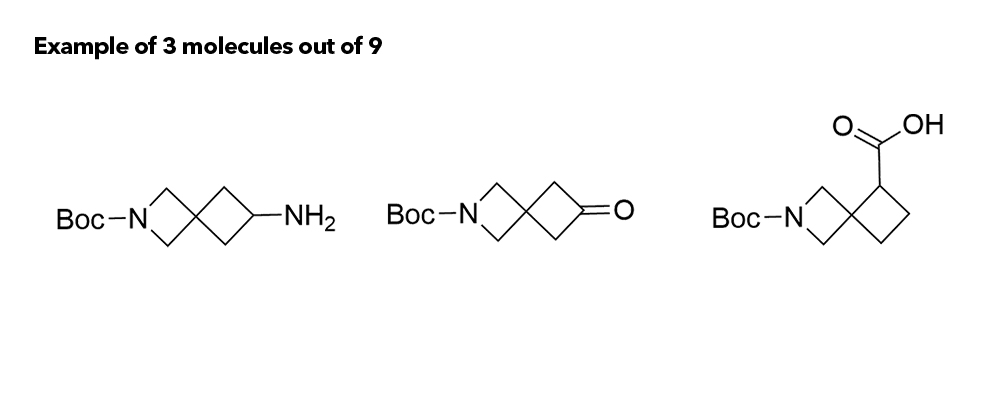
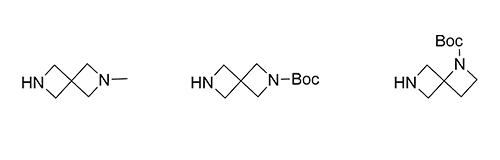
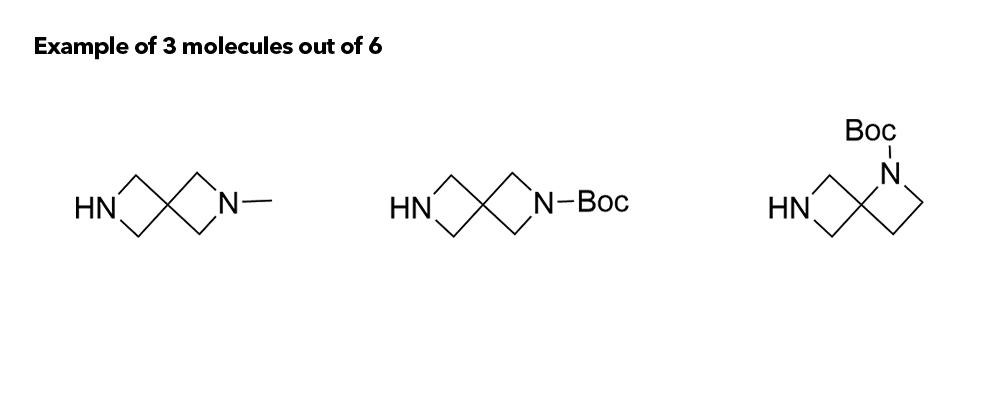
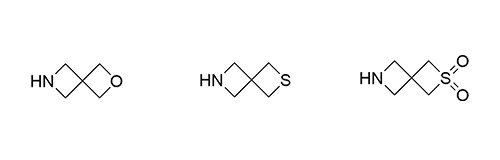
1b Synthesis of Azaspirocycles and their Evaluation in Drug Discovery
Angew. Chem. Int. Ed. 2010, 49, 3524
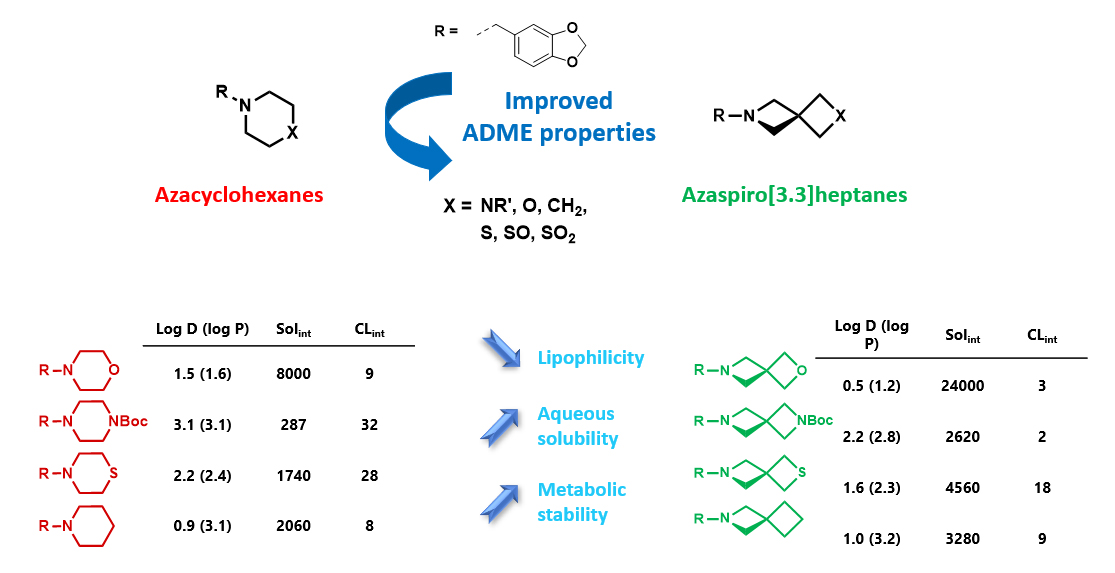
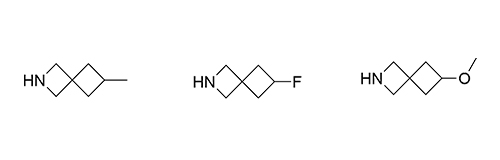
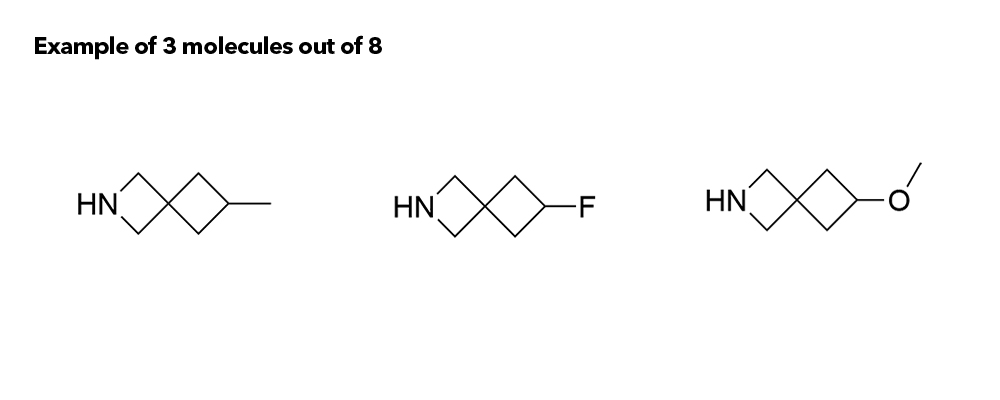
Although metabolic susceptibility is dependent on the structural context, and thus cannot generally be attributed on a given structural subunit of interest, it was found that most spiro[3.3]heptane derivatives were oxidatively degraded at lower rates than the six-membered monocyclic analogues in both human and mouse liver microsomes.
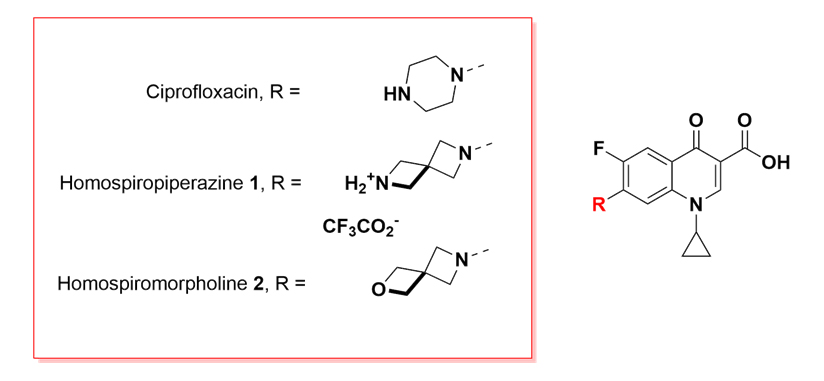
As case study, the homospiropiperazine and homospiromorpholine analogs of the antibacterial compound ciprofloxacin were prepared and evaluated.
The homospiropiperazine derivative 1 displayed 4-8-fold weaker inhibition than ciprofloxacin, however the homospiromorpholine analog 2 displayed comparable activity.
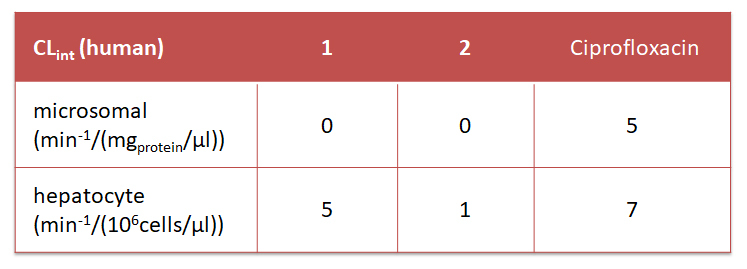
Moreover, metabolic studies revealed spiroisosters 1 and 2 to have high stabilities, with no observable metabolism in human microsomal assays whereas ciprofloxacin trifluoroacetate shows slight metabolic degradation. Likewise, 1 was as stable as ciprofloxacin in a human hepatocyte assay, whereas the homospiromorpholine analogue 2 remained essentially unaffected under the same whole-cell assay conditions.







![Azaspiro[3.3]heptane derivatives](https://spirochem.com/themes/kara5/assets/img/btn-down.svg)